WASHINGTON, DC — VA failed to provide many COVID-19 patients with all the required information when prescribing them remdesivir, according to a recent VA Office of the Inspector General (OIG) report. Veterans were not told about side effects, that the drug was not Food and Drug Administration-approved at the time or that they had an option to refuse the treatment, the investigators pointed out.
They also found that VA failed to report nearly a quarter of the adverse events that patients experienced to the FDA. OIG officials said they are concerned that, although remdesivir is now fully approved, similar lapses could occur with future emergency-use medications.
On May 1, 2020, the FDA authorized the emergency use of the investigational antiviral remdesivir to treat severe COVID patients. The FDA provided information on specific laboratory tests to be ordered prior to and during the administration of the drug to track its effectiveness, and required all adverse events be reported within seven days of occurring.
One week later, VA issued a memorandum outlining the use of the drug under FDA’s emergency use authorization (EUA) criteria. Because of its short supply and specific storage requirements—the prepared solution remains stable in only for a limited time—VA needed someone available around the clock to accept overnight, cold-chain shipments of the drug and to report any unused medication to VA’s Emergency Pharmacy Services group.
In August 2020, the FDA amended its EUA to include all hospitalized adult COVID patients. And in October 2020 the agency fully approved the drug for use in all adults.
OIG investigators looked at the health records of 825 VA patients administered remdesivir under the EUA from May 8 through Oct. 21.
They found multiple deficiencies with patient or caregiver education and the timely report of adverse events to the FDA.
OIG determined that VA did not:
- provide 62% of patients or caregivers with the fact sheet about the drug;
- inform 41% that remdesivir was not an FDA-approved mediation;
- inform 37% of the risks and benefits of the drugs;
- advise 59% that there were alternatives to the drug; and
- notify 32% that they had the option to refuse the medication.
“This could have resulted in patients or caregivers lacking the information needed to make a fully informed decision to receive the medication,” OIG states in its report.
According to VA leaders interviewed for the report, providers likely educated patients but failed to document it.
Common side effects associated with remdesivir include nausea and mild elevations in liver enzyme levels. The more serious side effects include infusion reactions, allergic reactions and kidney injury.
The report also found that VA failed to report 10 of the 42 adverse events that occurred in that sample of 825 patients to the FDA. Another six of the events were reported later than the required seven days. FDA requires this so that it can evaluate the safety of the drug in as close to real time as possible.
The OIG made no recommendations about remdesivir specifically, but rather suggested that VA look at its administration of EUA medications as a whole and better prioritize patient communication and safety.
Whether remdesivir actually improves patient outcomes remains in question. Study findings have swung back and forth between the antiviral providing a small benefit to it providing none at all, or even having a negative impact by lengthening hospital stays.
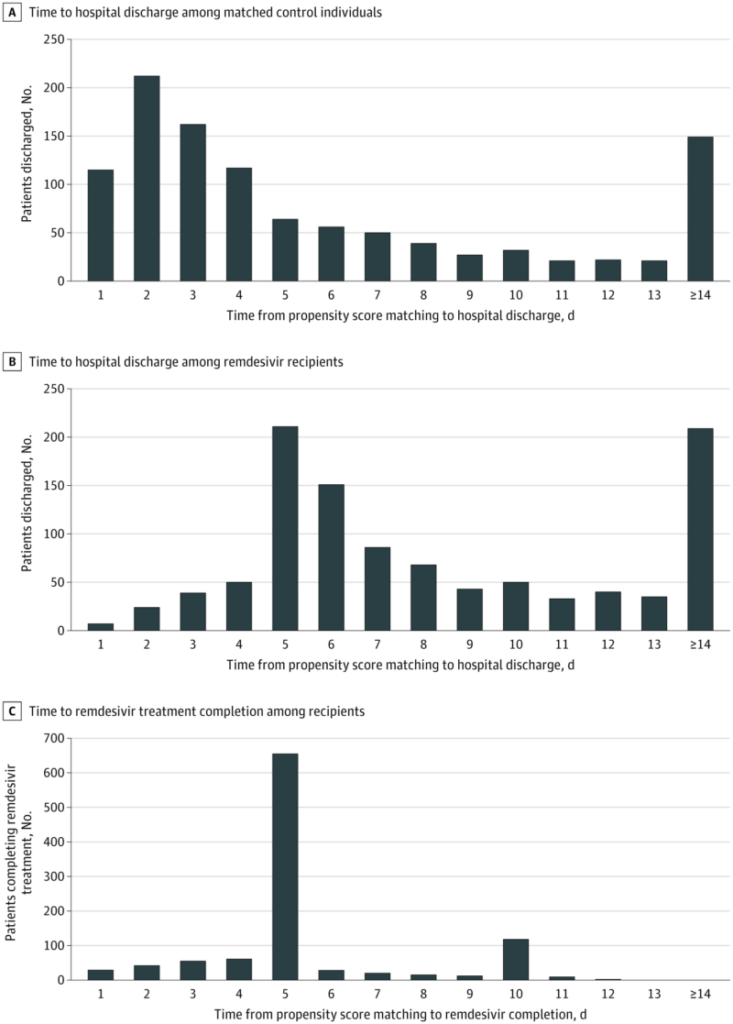
A VA study released in July 2021 found that remdesivir was not associated with improved survival for COVID patients, and the study’s authors found that clinicians had been keeping people in the hospital past when they needed to in order to complete the five- and 10-day remdesivir courses.1
A VA-funded review of studies released through October 2021 and published in May 2022 found that, in hospitalized adults, “remdesivir probably results in little to no difference in mortality.”
The report added, “Remdesivir may reduce time to clinical improvement and may lead to small reductions in serious adverse events, but may result in a small increase in any adverse event.”
- Ohl ME, Miller DR, Lund BC, et al. Association of Remdesivir Treatment With Survival and Length of Hospital Stay Among US Veterans Hospitalized With COVID-19. JAMA Netw Open. 2021;4(7):e2114741. doi:10.1001/jamanetworkopen.2021.14741