VA Treats As Much As 17% of All Parkinson’s in the United States

Changes in cognitive status across visits.
The number inside each node represents the number of people with the corresponding cognitive status indicated by its color. The nodes with dashed line represent people with only data from the first visit. The links represents the group participants who continued to the next visit.
PORTLAND, OR — The common image of Parkinson’s disease focuses on tremors and unstable movement, but for many patients with the disorder, the possibility of dementia worries them more. The concern is well justified—about 30% of patients with Parkinson’s disease (PD) suffer from dementia.
In recent years, researchers have sought to identify which individuals with PD have the greatest risk of dementia as a first step in targeting interventions that may prevent or delay its onset. That’s good news for the more than 110,000 veterans who have PD for whom identification of risk factors and successful therapies could preserve their autonomy and overall quality of life for additional years.
Veterans account for between 10% and 17% of all cases of PD in the United States, as a result of greater exposure to factors that increase the likelihood of developing the disease. Vietnam veterans who develop Parkinson’s disease have a presumed service connection because of the documented association with Agent Orange. Similarly, individuals based at Camp Lejeune from 1953 to 1987 have presumptive coverage for PD due to contaminants in the water supply. Traumatic brain injury, the signature injury of the conflicts in Iraq and Afghanistan, also increases the risk of PD.
Consequently, the VA developed six specialized Parkinson’s Disease Research, Education and Clinical Centers (PADRECCs) based in Houston, Philadelphia, San Francisco, Seattle/Portland, West Los Angeles and Richmond, Virginia. Each center provides state-of-the-art care to patients in its geographical region and conducts research, education and advocacy. In addition, the National VA Parkinson’s Disease Consortium extends the reach of PADRECCs with experts across the country.
Several VA researchers at the Northwest PADRECC, which is a collaborative effort of the VA Portland Health Care System and the VA Puget Sound Health Care System in Seattle, recently joined forces with investigators at Stanford University of Medicine in Palo Alto, CA, and others to develop a model that could predict dementia in patients with PD.1
It’s a challenging undertaking. Some patients with PD never develop cognitive issues; some have dementia at diagnosis. Others may have mild cognitive impairment for years, while yet another group of patients may have only brief impairment.
Several previous studies have examined the role of specific genes and certain phenotypes in the development of Parkinson’s disease dementia (PDD), but the disease and its cognitive manifestations have demonstrated remarkable heterogeneity. As a result, the authors noted that “[t]raditional statistical methods aimed at the identification of factors associated with cognitive progression may produce biased or spurious results.”
Developing a Better Model
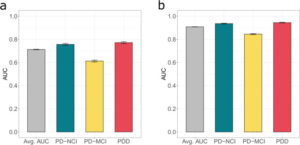
Biological factors satisfactorily predict cognitive status.
Cross-validated area under receiver operating characteristic (AUC) of the mixed-effect model prediction based only on biological factors (a) compared to the AUC of the mixed-effect model prediction based solely on cognitive tests (b). Error bars represent standard deviations (sd).
The VA researchers and their colleagues instead took a broad look at possible genetic, cognitive and clinical factors to craft a more useful, multivariate predictive model.
They analyzed data from 827 participants with PD enrolled in the Pacific Udall Center, one of nine Morris K. Udall Centers of Excellence for Parkinson’s disease research funded by the National Institutes of Health (NIH) to research causes of and treatment for PD. The Pacific Udall Center collects and deeply annotates longitudinal data from individuals with PD at Stanford University, the University of Washington/VA Puget Sound Health Care System and Oregon Health & Science University/Portland VAMC.
The team evaluated age, education, sex, time since onset of motor symptoms, length of follow-up, depression, total levodopa equivalent daily dose and site as covariates along with mutations in the APOE, GBA and MAPT genes and performance on the Unified Parkinson’s Disease Rating Scale. They also examined results of multiple individual cognitive tests including the Montreal Cognitive Assessment, Hopkins Verbal Learning Test-Revised (HVLT-R), phonemic verbal fluency, semantic verbal fluency, letter number sequencing and others.
They found sex, GBA carrier status, age and disease duration were the most important biological factors in progression to dementia. GBA mutations, the most common genetic risk factor for the disease, appear in 5% to 10% of all people with PD. Most individuals with PD have only one copy of the mutated gene, making them carriers. Two copies of the mutation cause Gaucher’s disease.
Overall, males were more likely to experience cognitive impairment and to decline more quickly. Male GBA carriers had the fastest progression to dementia, while female carriers progressed at about the same rate as males without the mutated gene. “Notably, the increase in odds ratios of both being male and having a glucocerebrosidase gene (GBA) variant were approximately equivalent to an additional 15 years of PD duration in terms of PDD risk in this cohort,” the researchers found.
On average, GBA carriers of both sexes performed worse in global function, divided attention, working memory, and processing speed. Male carriers also had reduced visuospatial function; females did not. Previous studies have linked visuospatial impairment with development of dementia.
All biological factors except MAPT and APOE genotype were significantly associated with cognitive status. Performance on the cognitive measures alone proved a more reliable indicator of future dementia or no impairment than the biological measures alone, however. Tests of executive function, semantic verbal fluency and recall in particular accurately predicted progression to PDD.
The multivariate analysis could reliably predict which patients would develop dementia and which would have no cognitive impairment. It did not effectively identify who would have mild cognitive impairment. The researchers suggested that mild cognitive impairment (MCI) can be fleeting, with 24% of individuals with MCI at one point having no cognitive impairment on later assessment. Medication, mood disorders and fluctuations in attention can affect cognitive evaluations and may change over time.
Throughout the study, the largest group of patients had mild cognitive impairments. Given the importance of this group and the difficulty in predicting which patients would fall in it, the authors called for additional research. They noted that “[f]uture work to better identify predictors of variability versus stability for those with PD-MCI will be important in the ongoing pursuit of optimally characterizing and introducing effective interventions for this sizable group of cognitively impaired individuals with PD.”
- Phongpreecha T, Cholerton B, Mata IF, Zabetian CP, Poston KL, Aghaeepour N, Tian L, Quinn JF, Chung KA, Hiller AL, Hu SC, Edwards KL, Montine TJ. Multivariate prediction of dementia in Parkinson’s disease. NPJ Parkinsons Dis. 2020 Aug 25;6:20. doi: 10.1038/s41531-020-00121-2.


