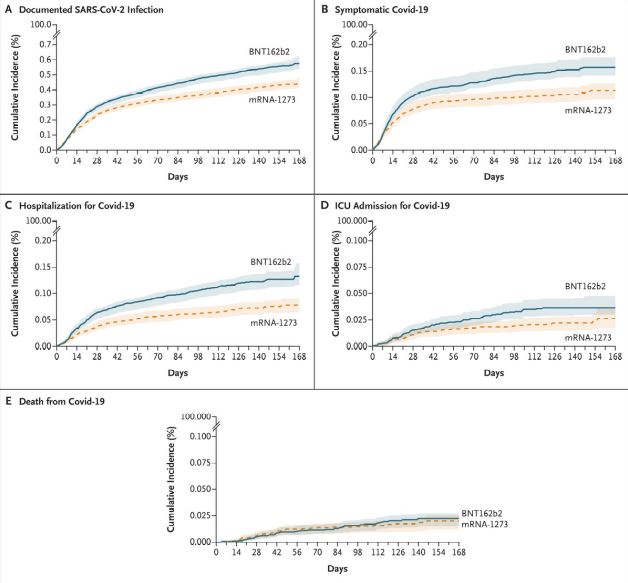
Click to Enlarge: Cumulative Incidence of Covid-19 Outcomes during a Period Marked by SARS-CoV-2 Alpha-Variant Predominance (January 4–July 1, 2021).
BOSTON — While both of the messenger RNA vaccines used in the United States work very well, a new VA study found that, in a head-to-head comparison, the Moderna product is more effective than the Pfizer-BioNTech product.
The report in the New England Journal of Medicine was based on an examination of U.S. military veterans who had received each vaccine. Overall, both vaccines were determined to be highly effective in preventing COVID-19 outcomes such as documented infection, hospitalization and death.
The Moderna vaccine, however, was found to offer an increased level of protection, including a 21% lower risk of documented infection and 41% lower risk of hospitalization, according to the study team led by researchers from the Harvard T.H. Chan School of Public Health, Brigham and Women’s Hospital and the VA.
Veterans receiving the BNT162b2 Pfizer-BioNTech vaccine were found to have a 27% higher risk of documented SARS-CoV-2 infection and a 70% higher risk of hospitalization for COVID-19, compared to recipients of the mRNA-1273 Moderna vaccine over 24 weeks of follow-up during alpha-variant predominance.
A higher risk of documented infection also was detected among recipients of BNT162b2 than among recipients of mRNA-1273 over 12 weeks of follow-up as the delta variant took over, but estimates are less precise because of the smaller number of eligible patients.
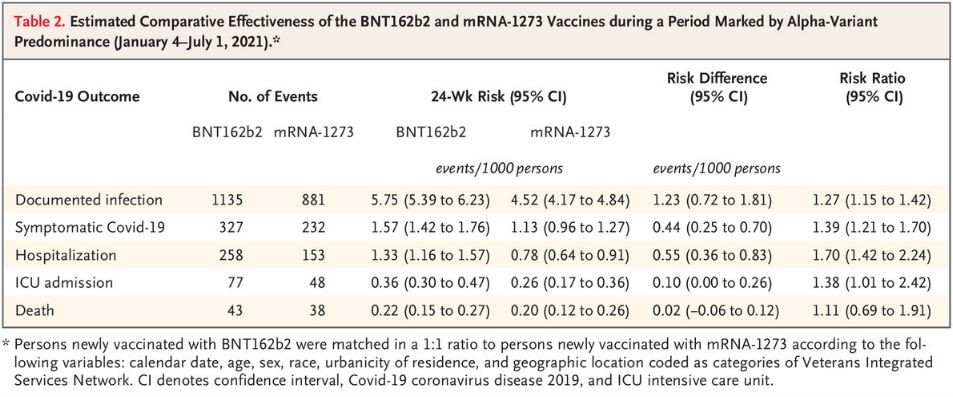
Click to Enlarge: Estimated Comparative Effectiveness of the BNT162b2 and mRNA-1273 Vaccines during a Period Marked by Alpha-Variant Predominance (January 4–July 1, 2021).*
“Both vaccines are incredibly effective, with only rare breakthrough cases,” said J.P. Casas, MD, PhD, an epidemiologist and associate professor with Brigham and Women’s Hospital and Harvard Medical School and executive director of the VA’s Million Veteran Program for genetics and health research. “But regardless of the predominant strain—Alpha earlier and then delta later—Moderna was shown to be slightly more effective.”
The comparative effectiveness study was designed to answer a persistent question: Which of the two mRNA vaccines is more effective. To determine that, effectiveness was measured with five COVID-19-related criteria:
- documented COVID-19,
- symptomatic disease,
- hospitalization,
- ICU admission, and
- death.
For the investigation, the authors used electronic health records of VA patients who received one of the two COVID-19 vaccines between early January 2021 and mid-May 2021. The first phase of the research focused on the Alpha variant that was predominant at the time. Recipients of the two vaccines—219,842 who got the Pfizer vaccine compared to the same number of Moderna recipients—were matched based on a range of clinical and demographic factors that could affect outcomes.
Results indicated that, over the study’s 24-week follow-up period, the estimated risk of documented infection was 4.52 events per 1,000 people in the Moderna vaccine group and 5.75 per 1,000 in the Pfizer group—an excess of 1.23 cases of documented infection per 1,000 people in the Pfizer group.
Smaller differences were with the other criteria: an excess of symptomatic COVID-19 (0.44 events), hospitalization (0.55 events), ICU admission (0.10 events) and death (0.02 events) per 1,000 people in the Pfizer group relative to the Moderna group.
Delta Variant Primary
Similar results were determined in an additional research phase during a time frame when the delta variant was the primary COVID-19 strain. Excess risk of documented infection over 12 weeks was 6.54 events per 1,000 people for the Pfizer vaccine, compared to Moderna. The supplementary research involved a shorter time frame, only analyzed infection as an outcome and was considered less precise because a smaller number of veterans were eligible for this analysis.
“Given the high effectiveness of both the Moderna and Pfizer vaccines, confirmed by our study, either one is recommended to any individual offered a choice between the two,” said the study’s first author, Barbra A. Dickerman, PhD, an epidemiology instructor with the Harvard T.H. Chan School of Public Health. “However, while the estimated differences in effectiveness were small on an absolute scale, they may be meaningful when considering the large population scale at which these vaccines are deployed. This information may be helpful for larger decision-making bodies.”
Researchers chose to do the research using the very large VA records system because, with millions of patients nationwide, the system allowed a very large sample size. That meant that even small differences in effectiveness between the Pfizer and Moderna vaccines could be pinpointed.
The VA databases also enable the study team to precisely characterize recipients of each vaccine type and closely match them on age, sex, race, geographic location and other attributes that could affect COVID-19-related outcomes.
“After this careful matching, we found that the two vaccine groups were extremely similar in terms of variables with respect to an extensive set of demographic, geographic and health-related attributes,” Dickerman said. “This allowed our observational analysis to produce exceptionally credible results during a global emergency, when answers are needed fast and randomized trials can be impractical.”
The authors pointed out that they “quantified the comparative effectiveness of the BNT162b2 and mRNA-1273 vaccines for the prevention of COVID-19 outcomes in the largest integrated health care system in the United States.”
“Our findings are consistent with those of studies that have reported a higher SARS-CoV-2–binding antibody response among recipients of the mRNA-1273 vaccine than among recipients of the BNT162b2 vaccine,” the authors wrote.
They pointed to an unpublished report that showed a lower risk of COVID-19 outcomes among recipients of mRNA-1273 than among recipients of BNT162b2 in the Mayo Clinic Health System (a study population that was approximately 95% white). “However, the interpretation of this finding is not straightforward, because the analyses were conditional on postbaseline factors (the study population was restricted to persons who underwent at least one SARS-CoV-2 PCR test during follow-up) and included few events for severe COVID-19 outcomes,” they wrote.
Another report which evaluated effectiveness against COVID-19 hospitalization compared the two vaccines with no vaccination in two case-control studies. “An indirect comparison of the results from the two case-control studies suggests that mRNA-1273 was more effective than BNT162b2 if the controls from both studies had similar characteristics, but the case–control design precluded estimation of absolute risk,” researchers wrote.
As to why the effectiveness might be different between the BNT162b2 and mRNA-1273 vaccines, researchers posited that could be the result of the following:
- different mRNA content of the vaccines (100 μg for mRNA-1273 vs. 30 μg for BNT162b2),
- the different interval between the priming and boosting doses (four weeks for mRNA-1273 vs. 3 weeks for BNT162b2), or
- other factors, such as the lipid composition of the nanoparticles used for packaging the mRNA content.
“Although this study provides evidence of potentially different effectiveness of the BNT162b2 and mRNA-1273 vaccines, any choice between two vaccines must also take their comparative safety into consideration, and safety was not studied here,” according to the article. “Head-to-head comparisons of the BNT162b2 and mRNA-1273 vaccines for safety outcomes are lacking, but early randomized trials identified only transient local and systemic reactions (e.g., pain at the injection site and headache) that are common among other viral vaccines, and observational studies and surveillance efforts have confirmed the safety of these vaccines for the population overall. In fact, even with respect to events for which the risk is increased after vaccination, the risk is even greater after natural infection with SARS-CoV-2 than after vaccination. Given the high effectiveness and the safety profile of both mRNA vaccines, either vaccine is strongly recommended.”
- Dickerman BA, Gerlovin H, Madenci AL, Kuransky KE, et. al. Comparative Effectiveness of BNT162b2 and mRNA-1273 Vaccines in U.S. Veterans. NEJM. December 1, 2021. DOI: 10.1056/NEJMoa2115463


