IOWA CITY, IA — Last October, the Food and Drug Administration approved remdesivir, marketed as Veklury, for use in adults and children 12 and older who weight at least 40 kg for the treatment of COVID-19 requiring hospitalization.
The FDA said the drug should only be administered in a hospital or in a healthcare setting capable of providing acute care comparable to inpatient hospital care.
Now, a VA study has joined others in raising questions about how effective remdesivir treatment really is in treating novel coronavirus. The report in JAMA Network Open sought to determine if the drug was associated with improved survival or shortened hospitalizations among people with COVID-19 in routine care settings.1
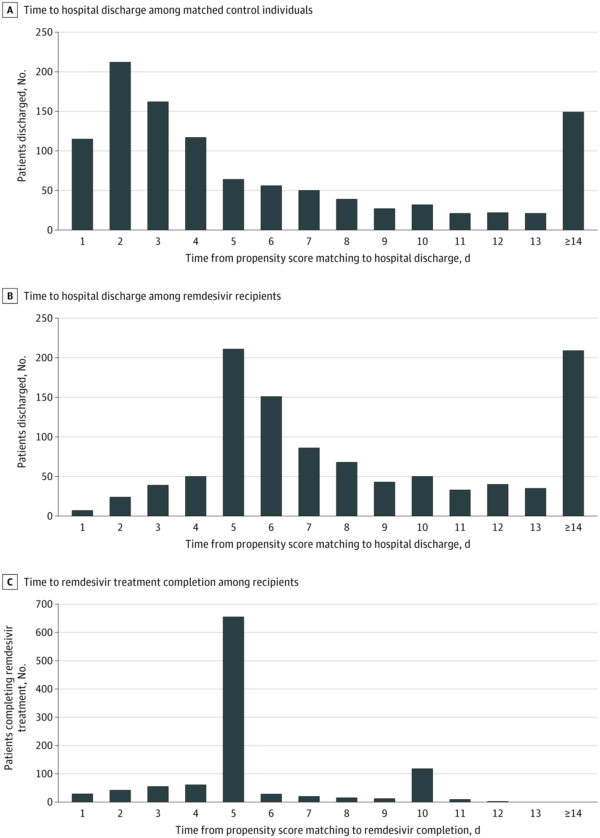
Researchers from the Iowa City VA Health Care System and colleagues conducted a cohort study of 2,344 US veterans hospitalized with COVID-19 to determine the answer. They found that remdesivir therapy was not associated with improved 30-day survival but was associated with a significant increase in median time to hospital discharge.
“The findings suggest that routine use of remdesivir may be associated with increased use of hospital beds but not with improvements in survival,” the study team advised.
The article noted, “Randomized clinical trials have yielded conflicting results about the effects of remdesivir therapy on survival and length of hospital stay among people with COVID-19.”
Their retrospective cohort study used data from the VHA to identify adult patients in 123 VMACs who had a first hospitalization with laboratory-confirmed COVID-19 from May 1 to Oct. 8, 2020. The authors compared patients initiating remdesivir treatment with control patients who had not initiated remdesivir treatment.
The focus was on time to death within 30 days of remdesivir treatment initiation (or corresponding hospital day for matched control individuals) and time to hospital discharge, with time to death as a competing event.
Of the initial cohort including 5,898 patients, 40.3%received remdesivir treatment, most, 94.3%, men with a mean age of 67.8. A majority, 59.7%, had never received remdesivir treatment before.
After propensity score matching, the analysis included 1,172 remdesivir recipients and 1,172 controls, leading to a final matched cohort of 2,344 individuals.
In addition to age and gender, remdesivir recipients and matched controls were similar with regard to dexamethasone use (559 [47.7%] vs. 559 [47.7%]), admission to the intensive care unit (242 [20.7%] vs 234 [19.1%]), and mechanical ventilation use (69 [5.9%] vs. 45 [3.8%]).
Results indicated that remdesivir treatment was not associated with 30-day mortality (143 remdesivir recipients [12.2%] vs 124 controls [10.6%]; log rank P = 0.26; adjusted hazard ratio [HR], 1.06; 95% CI, 0.83-1.36).
The authors pointed out that results were similar for people receiving vs. not receiving dexamethasone at remdesivir initiation (dexamethasone recipients: adjusted HR, 0.93; 95% CI, 0.64-1.35; nonrecipients: adjusted HR, 1.19; 95% CI, 0.84-1.69). Raising concerns among researchers was that remdesivir recipients had a longer median time to hospital discharge compared with matched controls (6 days [interquartile range, 4-12 days] vs. 3 days [interquartile range, 1-7 days]; P < 0.001).
“In this cohort study of U.S. veterans hospitalized with COVID-19, remdesivir treatment was not associated with improved survival but was associated with longer hospital stays,” the researchers concluded. “Routine use of remdesivir may be associated with increased use of hospital beds, while not being associated with improvements in survival.”
Background information in the articles explained that remdesivir (GS-5734) is a prodrug of an inhibitor of the SARS-CoV-2 RNA-dependent RNA polymerase and was one of the first drugs studied for treatment of people with COVID-19.
Conflicting Recommendations
The authors added, however, “Randomized clinical trials have produced conflicting results about the efficacy of remdesivir. The Adaptive COVID-19 Treatment Trial (ACTT-1) found that remdesivir shortened the time to illness recovery from a median of 15 days to 10 days among patients hospitalized with COVID-19. Remdesivir treatment in ACTT-1 was not associated with a reduction in mortality at 28 days (11.4% vs. 15.2%; hazard ratio [HR], 0.73; 95% CI, 0.52-1.03). The World Health Organization Solidarity Trial found that remdesivir treatment did not reduce the length of hospital stay or improve survival compared with the standard of care (rate ratio for death by 28 days, 0.95; 95% CI, 0.81-1.11). Other trials of remdesivir with varying designs have yielded equivocal results.”
Because of trial results have varied so much, conflicting recommendations regarding remdesivir use have been received by clinicians.
In addition to the FDA approval, the Infectious Diseases Society of America and the National Institutes of Health treatment guidelines currently recommend remdesivir treatment for people hospitalized with severe COVID-19. “These recommendations are partly based on the belief that if remdesivir use can shorten recovery time, it may allow more rapid discharge of patients from hospitals and open scarce beds to treat more patients during the pandemic,” the authors noted. “In contrast, the World Health Organization COVID-19 guidelines emphasize the lack of a survival benefit associated with remdesivir and recommend against the use of remdesivir for hospitalized patients.”
At the VHA, which is the largest integrated healthcare system in the United States, caring for more than six million veterans as of 2019. The VHA Pharmacy Benefits Management (PBM) created a centralized system to distribute remdesivir to VHA hospitals nationwide after the FDA issued an EUA and before it granted approval.
As of Oct. 1, 2020, VHA PBM had distributed remdesivir to treat more than 2,500 patients with COVID-19, according to the authors, which allowed them to study outcomes of remdesivir treatment in practice.
Their observational study combined PBM data on remdesivir distribution under the EUA with national VHA electronic records and administrative data to conduct a cohort study of the outcomes associated with remdesivir treatment among patients hospitalized with COVID-19.
In addition to studies like those conducted by VA researchers, other questions have arisen about the value of remdesivir.
A few months after the remdesivir approval, a study was published in the New England Journal of Medicine finding that the JAK inhibitor baricitinib plus remdesivir was superior to remdesivir alone in reducing recovery time. That study advised that the combination accelerated improvement in clinical status among patients with COVID-19, especially among those receiving high-flow oxygen or noninvasive ventilation, and did so with fewer serious, adverse events. Based on that information, the FDA issue an emergency use authorization for the drug combo to treat SARS-CoV2 infection.2
Fast-forward to late July, and the FDA revised the EUA, stating that baricitinib, marketed as Olumiant, now is authorized to be used alone for the treatment of COVID-19 in hospitalized adults and pediatric patients two years of age or older requiring supplemental oxygen, noninvasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO). In other words, the EUA for baricitinib no longer requires baricitinib be used in combination with remdesivir.
FDA officials explained that the EUA revision was supported by the COV-BARRIER clinical trial of hospitalized patients with COVID-19; in that study, baricitinib use appeared to reduce the proportion of patients who died through 28 days of follow-up compared to patients treated with the standard of care for COVID-19 alone. This study did not require baricitinib to be used in combination with remdesivir, and most patients didn’t get the second drug.
- Ohl ME, Miller DR, Lund BC, et al. Association of Remdesivir Treatment With Survival and Length of Hospital Stay Among US Veterans Hospitalized With COVID-19. JAMA Netw Open. 2021;4(7):e2114741. doi:10.1001/jamanetworkopen.2021.14741.
- Kalil AC, Patterson TF, Mehta AK, Tomashek KM, et. al. ACTT-2 Study Group Members. Baricitinib plus Remdesivir for Hospitalized Adults with Covid-19. N Engl J Med. 2021 Mar 4;384(9):795-807. doi: 10.1056/NEJMoa2031994. Epub 2020 Dec 11. PMID: 33306283; PMCID: PMC7745180.
- Marconi VC, Athimalaipet VR, de Bono S, Kartman CE, et. al. Baricitinib plus Standard of Care for Hospitalized Adults with COVID-19. medRxiv 2021.04.30.21255934; doi: https://doi.org/10.1101/2021.04.30.21255934