PHILADELPHIA — More than 5% of U.S. veterans are infected with chronic hepatitis C (HCV)—roughly triple the rate of the general population, with the percentage, 10%, even higher among those born between 1945 and 1965.
The comparable prevalence of HCV infection in the baby boomer cohort in the U.S. general population is much lower, at 2.4.
With the disproportionately high rate of HCV in the veteran population, a cure for the virus is a priority for the VA. Still, the VHA continues to treat about 174,000 of veterans with HCV.
The development of new hepatitis C treatments, especially the protease inhibitor-based direct-acting antivirals (DAAs), has been key for meeting that goal. A study last fall noted that VHA, the largest integrated healthcare system in the United States, provides unrestricted access to HCV treatments, and about 85% of its caseload has achieved cure.”1
But even as these drugs have increased HCV survival rates, concerns have been raised about their safety in some populations. Specifically, cases of liver injury have been reported in patients treated with protease inhibitor-based DAAs, but it is not clear whether risk of liver injury among people starting these drugs is increased compared to those initiating nonprotease inhibitor-based therapy. A new study led by researchers at Corporal Michael J. Crescenz VA Medical Center in Philadelphia offers some reassuring answers.2
“[FDA] warnings were based only on case reports, often occurring in people who had pre-existing severe liver disease, in whom these drugs are contraindicated,” explained lead author Jessie Torgersen, MD, an infectious disease physician at the Philadelphia VAMC and Instructor of Medicine at the University of Pennsylvania. The warnings raised questions about quantifying the actual risk of such events occurring from a population level, but no analyses have evaluated if initiators of PI versus non-PI-based DAAs have higher risk of acute liver injury (ALI), by advanced hepatic fibrosis/cirrhosis, according to the report.
The new study compared the risk of three ALI outcomes—alanine aminotransferase (ALT), severe hepatic dysfunction (coagulopathy with hyperbilirubinemia) and hepatic decompensation—in a cohort of 18,498 initiators of PI-based DAA therapy (paritaprevir/ritonavir/ombitasvir +/- dasabuvir, elbasvir/grazoprevir, glecaprevir/pibrentasvir) matched 1:1 on propensity score to non-PI-based DAA initiators (sofosbuvir/ledipasvir, sofosbuvir/velpatasvir) in the 1945-1965 Veterans Birth Cohort (2014-2019).
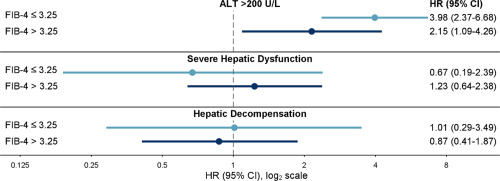
Researchers used Cox regression to determine hazard ratios (HRs) with 95% confidence intervals of each ALI outcome in PI versus non-PI initiators within groups defined by baseline Fibrosis-4 Index for Hepatic Fibrosis (FIB-4), noting FIB-4 >3.25 identifies people with advanced hepatic fibrosis/cirrhosis.
They found that, among veterans with baseline FIB-4 ≤3.25, PI initiators had a higher risk of ALT >200 U/L but not severe hepatic dysfunction or hepatic decompensation compared with non-PI-initiators. For those with baseline FIB-4 >3.25, PI initiators had higher risk of ALT >200 U/L but not severe hepatic dysfunction or hepatic decompensation compared to non-PI initiators.
Liver Inflammation
“Overall, the risk of acute liver injury was low, less than 1.5%,” Torgersen told U.S. Medicine. “We found that people treated with protease inhibitor-based DAAs had approximately 2-4 times higher risk of liver inflammation noted on their laboratory tests when compared to people treated with nonprotease inhibitor-based DAAs. This increased risk was observed in people with and without suggestion of advanced liver fibrosis or cirrhosis. However, there was no increased risk of severe hepatic dysfunction and liver decompensation in people taking protease inhibitor-based DAAs when compared to people treated with non-protease inhibitor based DAAs.”
While the study showed liver aminotransferase elevations occurred more commonly with protease inhibitor-based DAA, risk for the more severe and clinically apparent acute severe hepatic dysfunction or hepatic decompensation was not increased. “These findings show there is comparable hepatic safety of protease inhibitor-based and non-protease inhibitor based DAA therapies for people without decompensated cirrhosis,” Torgersen said.
The findings are reassuring for these drugs, which are widely used by the VA, she pointed out. “In our study, we found 28% of patients were treated with protease inhibitor-based DAAs,” she said. “Currently, elbasvir/grazoprevir and glecaprevir/pibrentasvir remain the first-line protease inhibitor-based DAAs on the VA formulary for hepatitis C cure.”
Before 2011, hepatitis C treatment was limited to interferon-based regimens, requiring weekly injections of medication, a protracted treatment course, and associated with substantial side effects and low cure rates, Torgersen said. The first generation of protease inhibitors included boceprevir and telaprevir. When used in combination with interferon, they increased cure rates to approximately 50%.
“While this represents a modest improvement in hepatitis C cure, it accelerated further development of novel hepatitis C treatments,” she said. IFN-free direct-acting antivirals became available shortly thereafter, with paritaprevir/ritonavir/ombitasvir/dasabuvir (PrOD) representing one of the first protease inhibitor-based DAA regimens that could be administered as all-oral treatment without the need for interferon, she said.
“Our findings reinforce our understanding that these hepatitis C treatments are safe and severe acute liver injury events, as those reported in the FDA’s warnings, are overall rare in people without decompensated cirrhosis,” Torgersen said. “Providers will likely be reassured with their use of PI-based DAAs in similar patients and be able to counsel their patient on the low risk of liver-related toxicity with these medications.”
- Beste LA, Green P, Berry K, Belperio P, Ioannou GN. Hepatitis C–Related Hepatocellular Carcinoma Incidence in the Veterans Health Administration After Introduction of Direct-Acting Antivirals. JAMA. 2020;324(10):1003–1005. doi:10.1001/jama.2020.10121
- Torgersen J, Newcomb CW, Carbonari DM, et al. Protease Inhibitor-based Direct-acting Antivirals Are Associated with Increased Risk of Aminotransferase Elevations but Not Hepatic Dysfunction or Decompensation. J. Hepatol. Published online July 28, 2021. DOI :https://doi.org/10.1016/j.jhep.2021.07.021